How TOPS Works
TOPS is Different from
Fusion—It Moves
Fusion eliminates all motion at the operative level.
TOPS preserves mobility of the spine segment.
Fusion places more stress and strain on the adjacent levels
TOPS protects neighboring segments.
Adjacent level disease or adjacent segment degeneration is a condition that can occur after a spinal fusion. It impacts the motion segments (i.e., the disc and facet joints) above and below the surgery site. After a fusion surgery, you lose movement at the spine segment where the procedure was done. But motion has to come from somewhere to accommodate your daily activity—twisting, walking, reaching, and lifting. Usually, it comes from the joints next to the surgery site. These joints have to do double duty in order to make up for the (now) immovable portion of your spine. As such they are subject to extra stress, and this may lead to degenerative changes. Adjacent level disease is the result of extra wear and tear on the intervertebral joints above and below your surgery site.
The TOPS™ System is a mechanical device that is housed between two titanium plates, which allows axial rotation, lateral bending, extension, and flexion. The implant facilitates bending, straightening and twisting movements at the affected segment of the spine while blocking excessive posterior and anterior sagittal translation.
Regain your mobility with Premia Spine! Contact us now
The TOPS™ System replaces anatomical structures, such as the lamina or the facet joint that are removed from the vertebrae during the spinal decompression treatment to alleviate pain. The internal metal stoppers replace the natural bony elements that served as stoppers during axial rotation. The boot and internal components take the place of the supraspinous ligament, interspinous ligament, and ligamentum flavum in their ability to help control flexion. The TOPS System is anchored to the spine with four standard polyaxial pedicle screws and is implanted via a traditional posterior surgical approach. The TOPS patented crossbar configuration exerts less force on the screws than fusion implant devices. At the same time, TOPS withstands sheer forces that interspinous and intralaminar devices cannot contain.
The TOPS™ System uniquely recreates motion in all directions – flexion, extension, lateral bending, and axial rotation. Clinically proven to provide immediate and sustained pain relief and improvement in quality of life, Premia Spine’s TOPS™ System has also been demonstrated to provide excellent outcomes for appropriate patients. The TOPS System provides three major benefits for the treatment of spinal stenosis. First, the procedure stabilizes the posterior spine and reestablishes a controlled range of movement. Second, patients regain their ability to bend, flex, walk, and enjoy the normal activities of life. Third, and most importantly, patients experience immediate and sustained pain relief. With these benefits, the device has earned a superiority-to-fusion claim from the FDA. Clinical studies conducted since 2005 show the TOPS System alleviates persistent leg and low back pain for patients with moderate to severe spinal stenosis, degenerative spondylolisthesis (a slipped disc), and facet arthrosis (bone spurs).
Getting the Motion You Deserve
Laboratory studies on the TOPS™ System have been performed at leading centers in the United States and Europe. A few of the studies are highlighted below.
The TOPS™ System allows full motion at the operative level after surgery. TOPS engineers invested over a year at the leading research laboratories of the world to design the TOPS. In Ulm, Germany ground-breaking research on the role of the facet joints was conducted in 2004. With the assistance of Professor Wilke, our engineers completed the design and testing of the TOPS device, successfully replicating the function of the facet joints. The culmination of the program was a landmark cadaver study published in Spine demonstrating TOPS’ ability to return a defective spine segment to normal motion.
Range of Motion: TOPS™ System versus destabilization
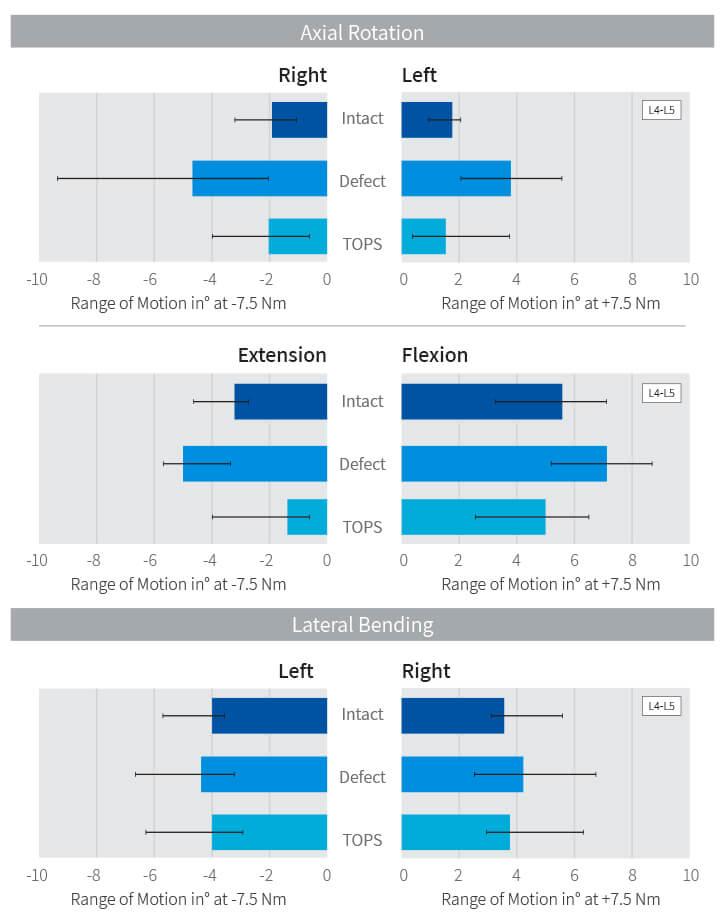

The company went on to examine the TOPS implant in the context of multi-level lumbar motion. The preclinical testing demonstrated that the TOPS™ System protects the adjacent levels. The experiment, conducted by Dr. Bryan Cunningham, called for cadaveric lumbar spine segments to each undergo the same motion four consecutive times. The contribution of each segment—L3-L4, L4-L5 and L5-S1—was measured each time. The first simulation was performed with intact spine specimens to establish baseline measurements. After destabilization of the L4-L5 segment, the global motion was repeated. Then the L4-L5 segment was fixed with screws and rods. Finally, the rigid fixation was replaced with TOPS. In each simulation, the contribution of each segment to the overall movement was measured.
The graph below shows that a destabilized segment moves over 60% more in axial rotation than before the infraction. The instability allows exaggerated unconstrained motion (and is therefore a source of pain). In this situation, the two adjacent segments move less as part of the global motion. In the third round with the semi-rigit simulation, the inverse is the result. With the fixation, the L4-L5 segment moves much less, forcing the adjacent segments to move more (i.e., work harder) in order to achieve the same degree of global motion. Finally, with the TOPS at L4-L5, the segments above and below the device move normally. It is posited that the levels adjacent to a fusion accelerate their normal degeneration process due to the added stress and strain that are lessened with TOPS.
Adjacent Level Range of Motion: TOPS™ System versus Fusion

One of the uniqueness of the TOPS is that the motion device is free floating with minimal loads exerted on the implant and its pedicle screws during common daily activities. At the extreme ranges of flexion, extension, lateral bending and axial rotation, peak loads are absorbed by the sophisticated biomechanical device. Unlike fusion or dynamic stabilization rod systems, the TOPS device links the pedicle screws at the same segment like a cross-connector (e.g., L4 to L4, and L5 to L5) instead of linking between adjacent segments (e.g., L4 to L5). With the TOPS, the segments interface via a flexible joint at their contact point. Loads are therefore shared across all four pedicle screws, the mobile implant, and two vertebral bodies. This is not the case with rigid fixation and dynamic rod systems. The result is significantly higher moments on the screws with more rigid systems. In addition, the variability in the loading creates a toggling effect that further weakens the screw-to-bone interface.
Clinical-biomechanical success with the TOPS™ System screws and arms has been demonstrated since January 2005 with a screw loosening incidence rate of less than 1% and no rod breakage in over 6,000 screws and rods worldwide.
Load on Pedicle Screws: TOPS System vs. Dynesys Stabilization System


Why trade-off adequate decompression with post-op stability
Most surgeons would agree that the one of the keys to a successful fusion surgery is the quality of the decompression. However, the optimal breadth of the decompression can be limited by concerns for leaving enough bone for fusion bridging. This is the tradeoff when performing fusion surgery.
With the TOPS™ System, clinicians can remove all elements pressing on nerve roots without concern for leaving enough bone for fusion to occur—because there is not fusion. Instead of permanently locking two adjacent vertebrae, clinicians allow the two vertebrae to continue moving normally with the TOPS™ implant stabilizing the segment and allowing motion at the operative level.
The TOPS™ System serves the function of the lamina and facet joints to prohibit excessive bending, straightening and twisting movements at the affected segment of the spine while blocking excessive posterior and anterior sagittal translation. No other non-fusion device can do this.
The result is optimum decompression without compromising stability. And if a revision of a TOPS became necessary, the locking caps are easily released and the TOPS device is removed. The surgeon can then rotate the screw heads and place two rods and a cage. A failed fusion procedure cannot be converted to a TOPS.
Clinical Outcomes with TOPS
Anekstein et al. published an 11-year follow-up study of 10 patients who were treated with the TOPS System. The primary indication for that study was neurogenic claudication due to spinal stenosis with single level degenerative spondylolisthesis. The study enrolled 5 males and 5 females aged 52-69 years (average 61.3 years). Outcome measures included VAS for leg and back pain, the Oswestry Disability Index (ODI) and the SF-36 health survey. These measures were administered preoperatively, and at 6 weeks, 3 months, 6 months, and at 1, 2, 3, 7 and 11 years.
There was one device-related failure 3 months post-surgery. X-rays showed that an internal component was damaged leading to a locking of the device. However, the screws were solidly anchored to the spine. After the 6-month follow-up, this patient underwent revision to replace the motion implant with fusion rods. No complications resulted from the fusion surgery. A minor design change in the prosthesis provided a solution to this rare complication, as confirmed by biomechanical testing of the alteration. The remaining 9 patients were seen at 1, 2, 3, 7 and 11-year follow-up.
Independent radiographic analysis at eleven (11) years revealed no evidence of spontaneous fusion or screw loosening or breakage. Flexion-extension and lateral bending views showed the TOPS implant to be mobile; and the patients demonstrated well-preserved motion clinically. MRI demonstrated no cases of stenosis or spondylolisthesis at the segment adjacent to the stabilize segment at 11 years, although progressive degeneration could be seen at 2, 3 and 4 levels above the index level in three patients.
All outcome measures demonstrated a statistically significant difference between pre and postoperative scores (p<0.05). The results are summarized in Table 1 below.


The TOPS System was clinically investigated in a randomized multi-center prospective clinical trial in the United States. The TOPS System was prospectively studied in a 1:1 randomization to traditional posterolateral fusion. Patients either received the TOPS device or a posterolateral fusion with either a DePuy (Johnson & Johnson) or a Stryker pedicle screw and fusion rod system. Patients were seen at 6 weeks, 3 months, 6 months, one year, and annually thereafter. Data collected included Oswestry Disability Index scores, VAS (pain) scores for leg and back, neurological assessments, and radiographic assessments.
The intent of this study was to complete the collection of clinical data on subjects enrolled in the TOPS IDE clinical trial (#G060063) sponsored by Impliant.
Six sites received IRB approval for continued follow-up and at those sites a total of 46 subjects who were enrolled returned for no less than two-year follow-up (out of a total of 54 subjects who were originally enrolled at six sites).
To the 2016 Annual Update, in the FDA report, 18 of the original 24 TOPS subjects remain in active follow-up and 12 of the original 21 Fusion subjects remain in active follow-up. All subjects have completed a minimum of five years of follow-up with many having additional long-term follow-up as follows:
• 6 Years – 17 subjects
• 7 Years – 15 subjects
• 8 Years – 6 subjects
• 9 Years – 2 subjects
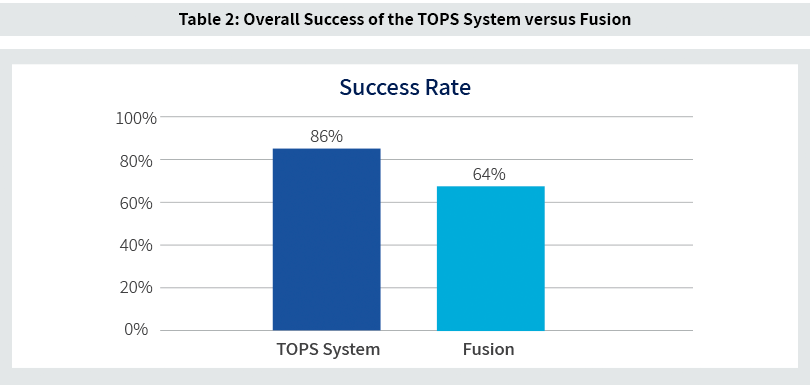
Clinical results as demonstrated by a composite clinical endpoint assessed at 24 months, or at next available time point, shows an advantage to the TOPS System versus fusion of 86% success rate among the TOPS patients versus 64% success rate among the fusion patients. The Success Rate was measured as a composite success, with each patient required to succeed in all the criteria below to be deemed a success. Missing any of the criteria categorized the patient as a failure:
• ODI improvement of at least 15 percent
• Maintenance or improvement in neurological status
• No radiographic device failure
• No radiographic screw loosening
• Radiographic absence (TOPS) or presence (Control) of fusion
• Freedom from additional surgical intervention defined as revision, non-elective removal or supplemental fixation
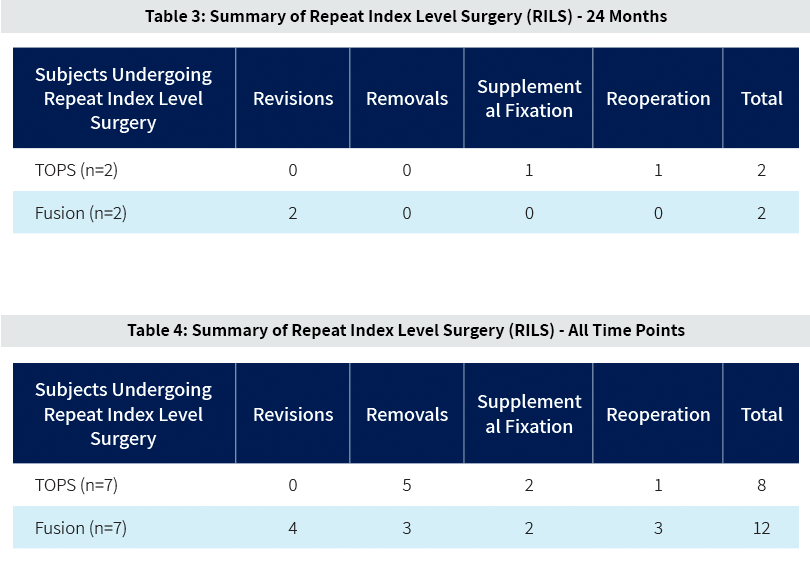
The incidence of repeat index level surgery (RILS) for both treatment groups is summarized below. Through 24 months the rate of RILS in the TOPS group was 8.3% and in the fusion group was 9.5% (see Table 3). It should be noted that the one reoperation occurring in the TOPS group was for a successful dural repair at 11 days post index surgery. Table 4 reports on RILS for all patients through all available time points.
Retrospective, Single-Center Study of 17 Patients Treated with TOPS
Dr. Werner Lack et al. have reported on 17 patients who were treated with TOPS at a single center in Vienna, Austria. The primary indication in these patients was spondylolisthesis with relative or absolute spinal canal stenosis (15 patients), lumbar spondylarthrosis with vertebrostenosis (1 patient), and severe facetarthrosis (1 patient). The sample of 17 patients included 10 female and 7 males with age range of 54 to 82 years old. Follow-up period at the time of the report was 26 – 77 months (51-month average).
All patients underwent decompressive laminotomy and facetecomy and 3 patients received a concomitant fusion at the neighboring segment. Additionally, the 14 patients whose primary diagnosis included a spondylolisthesis underwent reduction prior to final implantation of the TOPS implant. Levels treated with TOPS included L4/5 (10 patients), L3/4 (6 patients), and L2/3 (1 patient). The average operative was 102 minutes (60 – 140).
Outcome measures assessed included:
• Operative Duration
• Intra-Operative Complications
• Post-Operative Complications
• VAS
• Maintenance of Spondylolisthesis reduction
A single intra-operative complication was reported, that being an uncomplicated dural leak. Post-operatively one patient was reported as having asymptomatic screw halo at 6 months, however this patient’s original index-level procedure included a change of the screw position which could have contributed to this radiographic finding.
The average visual analog score (VAS) reduction was 7.5 at 3 months, 6.8 at 12 months, and 6.7 at the longest follow-up (average: 51 months, range: 26-77), demonstrating an average improvement of 81%. The preoperative and postoperative average ROMs were 8.2° and 7.4°, respectively.
Prospective, Single Center, Non-Randomized Study of 11 Patients Treated with TOPS
Haleem et al. have reported on 11 patients treated with TOPS at University Hospital Southampton in the United Kingdom. Primary indication for inclusion into the study was at least moderate stenosis with a Grade I spondylolisthesis. Primary exclusion criteria included previous lumbar fusion surgery, a spondylolisthesis of Grade II or greater, and any history of metabolic bone disease.
The study enrolled 11 patients however baseline evaluations for 1 patient were misplaced (this patient had successful results after surgery) and as such 10 patients are included in the analysis. These included 2 male and 8 females with an average age of 61 (range 52 – 73). All patients were Caucasian with average BMI of 28 (range 21 – 36).
Outcome measures included:
• Weight bearing AP and Lateral radiograph
• Magnetic Resonance Induction (MRI) scans – Lumbar
• Visual analog scale (VAS) for back and leg pain
• Oswestry Disability Index (ODI) Questionnaire
• SF-36 Survey
• Incidence or repeat index level surgery (RILS)
Patients were seen pre-operatively and then at 6 weeks, 3 months, 6 months, 1 year, 2 years and at the last visit—which for the cohort was an average of 5 years. The range follow-up at the final visit was 55 – 74 months, with none being lost to follow-up.
Patient reported outcomes (ODI, VAS, SF-12) all showed dramatic improvement at the first post-op time point and these improvements were maintained. The charts below summarize these data through an average 5-year follow-up.

In addition to the above, the investigators reported that none of the 10 patients included in the analysis underwent repeat index level surgery and no screw loosening was observed.
Prior to the FDA’s approval of the TOPS™ device in June, 2023, Premia Spine completed the enrollment of an FDA-approved investigation of the TOPS System in 300 patients at 37 investigational centers in the United States. Patients were randomized into either the TOPS (200 patients) or fusion arm (100 patients) of the study. The study is currently in the follow-up phase. During the clinical trial phase, the FDA designated the TOPS System as a Breakthrough Device Category, and Medicare (CMS) granted TOPS a Category B pre-approval for the coverage of routine care and services, including the device, furnished to Medicare beneficiaries enrolled into this trial.
A Prospective Study of Lumbar Facet Arthroplasty in the Treatment of Degenerative Spondylolisthesis and Stenosis: Early Cost-effective Assessment from the Total Posterior Spine (TOPS™) IDE Study.
Ament et al. reviewed the data from the FDA study to calculate the potential healthcare and societal benefits of the TOPS device versus lumbar fusion. Specifically, the study explored the cost-effectiveness and economic analysis of the TOPS System versus TLIF for patients suffering from spondylolisthesis and spinal stenosis. Patients were randomly assigned to either TOPS or TLIF surgery as part of a prospective, multi-center U.S. Food and Drug Administration (FDA) clinical trial.
The data from the study was analyzed by Neuronomics, an independent professional corporation focusing on healthcare economic analyses. Its findings demonstrate that, even at a premium of $4,000 over the cost of a TLIF implant, TOPS still achieves cost-effectiveness at a willingness-to-pay threshold of $100,000 within one-year after surgery. Furthermore, TOPS becomes the dominant strategy when data is examined at two years and beyond. From the societal perspective, TOPS is even more highly cost-effective at one year and dominant at two years and beyond.
TOPS appears to be a highly cost-effective surgical modality compared with TLIF as a motion-preserving, non-fusion alternative for the treatment of grade 1 spondylolisthesis with lumbar spinal stenosis, with the TOPS System yielding greater quality of life at a lower total cost over time.








